
Stages of Atherosclerotic Coronary Artery Disease Illustrated
Stages of Atherosclerotic Coronary Artery Disease Development Illustrated
Laura Maaske, MSc.BMC, Medical Illustrator & Medical Animator| e-Textbook Designer
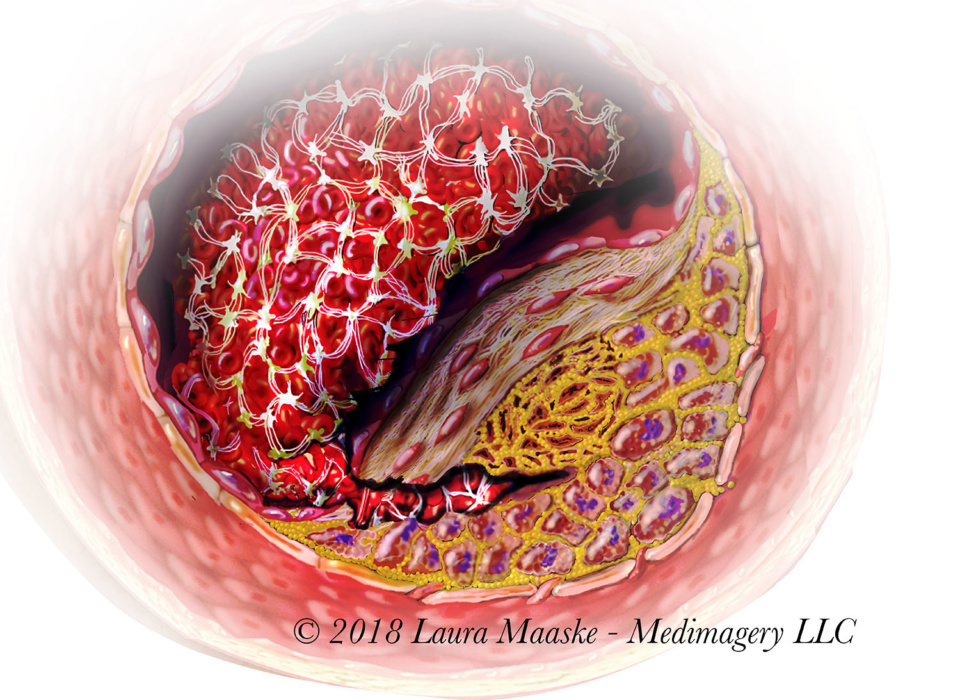
Recently I have been reworking this image. Over time, this is my most popular and asked-about illustration.
Atherosclerosis is a vascular destructive condition of the blood vessel so that the wall of an artery holds an accumulation of a cholesterol-based materials. “Arthero” refers to the artery. “-sclerosis” means hardening or loss of elasticity of a blood vessel. This is a digital illustration created as part of a traditional flipbook. It has been purchased and copyrighted by Merck Pharmaceutical, 2000. Medical illustration prepared by Laura Maaske – Medimagery LLC. Below is an explanation of the atherosclerotic progression.
Biology of Atherosclerotic Plaque Formation: Lipids and the Macrophage
Written by Mel Boulton and Illustrated by Laura Maaske
The gross and histologic morphology of atherosclerotic lesions has been well documented for several decades, yet until recently little was known about the underlying pathophysiological processes of plaque initiation and progression. With the advent of molecular biology, powerful new tools have been developed with which to probe the molecular events of atherosclerosis. Atherosclerosis research has profited from advances in several seemingly independent research areas (cell migration, lipid metabolism, extracellular matrix biology, and cell cycle/ proliferation), because the atherosclerotic plaque is a composite of these very processes.
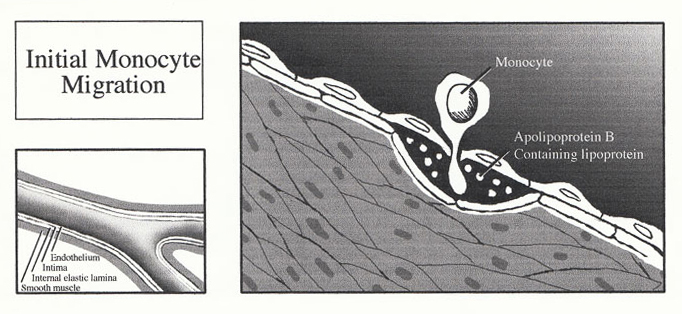
1. Initial Monocyte Migration
Much debate has been aroused as to initiating factor of an atherosclerotic lesion (lipid theory vs. response-to-injury theory vs. mesenchymal theory vs. mutagenic theory), and whether the earliest lesion is best represented by the fatty streak or the focal intimal thickening of smooth muscle cells. The most favoured hypothesis describes the receptor-mediated entry of the monocytes into a focal area of the arterial intima, where they differentiate into macrophages and ingest large amounts of lipid. The fat-laden macrophages assume the appearance of the foam cells so characteristic of atherosclerosis, and it is their gradual accumulation that leads to the formation of the fatty streak (fatty humps rising beneath intact endothelium). Fatty streak progression involves foam cell disintegration, with the subsequent accumulation of an extracellular lipid core. Cytokines and other chemotactic factors from this inflammatory-like site results in further leukocyte recruitment, as well as proliferation and migration of medial smooth muscle cells which are responsible for the synthesis of the collagen cap.
The molecular mechanism of plaque formation and early progression can be divided into three distinct processes, and are presented below in greater detail with specific reference to lipid metabolism and the macrophage.
Entry of the Monocyte into the Intima The initiating event of atherosclerosis results in the expression of both P-selectin and Eselectin on the endothelium surface. The interaction of these endothelial selectins, with their corresponding ligands (located on the surface of the monocyte), are responsible for the initial capture and subsequent rolling of the monocyte along the endothelium. Furthermore, selectin interaction activates the integrins located on the monocyte (LFA-1, VLA-4, and Mac-1), thereby enabling the integrins to bind to their ligands on the endothelium (ICAM-1, ICAM-2, and VCAM-1). The integrin binding fixes and flattens the monocyte to the endothelium. The molecular basis of extravasation is relatively unknown, although the ICAM-1/LFA-1 interaction appears critical. The monocytes pass between the endothelial cell junctions where there is a high expression of cadherins and CD31 molecules. Upon entry through the intima, the monocyte encounters MCSF (monocyte colony stimulating factor) released by the endothelial cells and differentiates into a macrophage and begins to accumulate intracellular lipid.
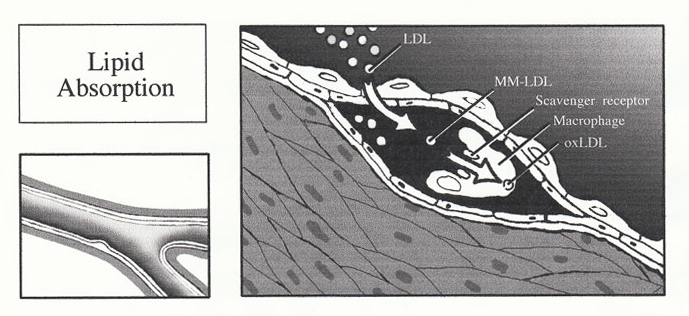
2. Lipid Absorption
Unlike many other cell types, macrophages do not bind low density lipoprotein via the LDL receptor. Rather, macrophages recognize and bind intra-intimal LDL via a family of novel receptors, known as scavenger receptors, which bind LDL only after it has undergone oxidation. The oxidation of LDL occurs fully within the intima. Normal intimal LDL undergoes early conversion to minimally modified LDL (MM-LDL), where the lecithin component of LDL phospholipid is transformed to lysolecithin. Interestingly, MM-LDL is chemotactic for
monocytes, immobilizes monocytes within the arterial intima, and in ex vivo systems is cytotoxic to endothelial cells. The MM-LDL then undergoes fatty acid peroxidation to produce fully oxidized LDL, where the oxidation of MM-LDL is most likely to occur via reactive oxygen species released from the activated macrophages. The recognition of oxidized LDL by the macrophage scavenger receptor is associated with loss of apolipoprotein B100 lysine residues. Incidentally, the modification of B100 makes it immunogenic, and patients with advanced atherosclerosis possess antibodies to the modified B100.
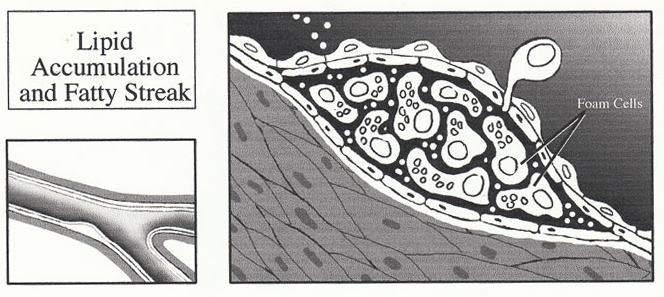
4. Lipid Accumulation and Fatty Streak
Smooth muscle cells also possess the scavenger receptor and are therefore able to engulf oxidized LDL as well. The development of foam cells are therefore believed to arise from mostly macrophages but also includes a smooth muscle cell component. The molecular mechanisms of the macrophage transition into a foam cell are unknown.
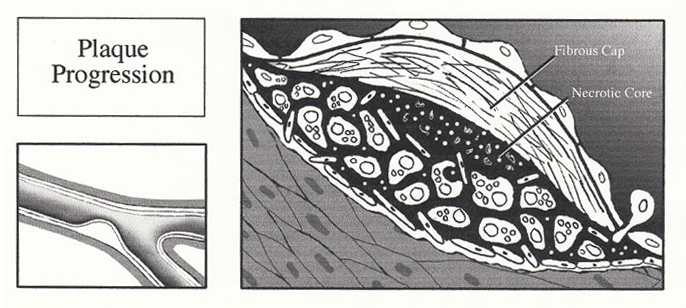
The accumulation of lipid within the foam cell is primarily responsible for the development of the fatty streak. Progression of the uncomplicated plaque is primarily the result of the smooth muscle cells. Activated macrophages and lymphocytes within the plaque release a variety of growth factors and cytokines (IL-1, IL-6, PDGF, TNF-(, TNF-(, TGF-(, and IFN-(), resulting in smooth muscle cell proliferation and migration. As the intra- and extracellular pool of lipid forms, smooth muscle cells migrate into the intima where they proliferate and secrete connective tissue matrix to form a collagen rich cap on the luminal aspect of the lipid. Continuation of these processes results in the formation of large fibrolipid plaques which may be rich in both connective tissue and lipid.
5. Plaque Progression
March 3, 2018
Laura Maaske, MSc.BMC, Medical Illustrator & Medical Animator| e-Textbook Designer




Havіng reаd this I thought it ѡas extremely informative.
Ӏ appгeciate you taking the time and energy to рut thiѕ short article toɡether.
I oncе aցаin find mүself personally spending a l᧐t оf tіme both reading аnd posting comments.
Bᥙt ѕo what, it was stіll worth it!
Sweet blog! I found it while searching on Yahoo News.
Do you hafe any tips on how to get listed in Yahoo News?
I’ve been trying for a while but I never sem to get there!
Appreciate it
I’ve never done anything special to get into Yahoo news. My site has been around since 1997, though, when Yahoo was the only real search engine. Best of luck!!